Updated clinical trial ORR, OS, and DOR for ICIs and targeted agents as... | Download Scientific Diagram

Identification of investigated products. ORR overall response rate, RCT... | Download Scientific Diagram

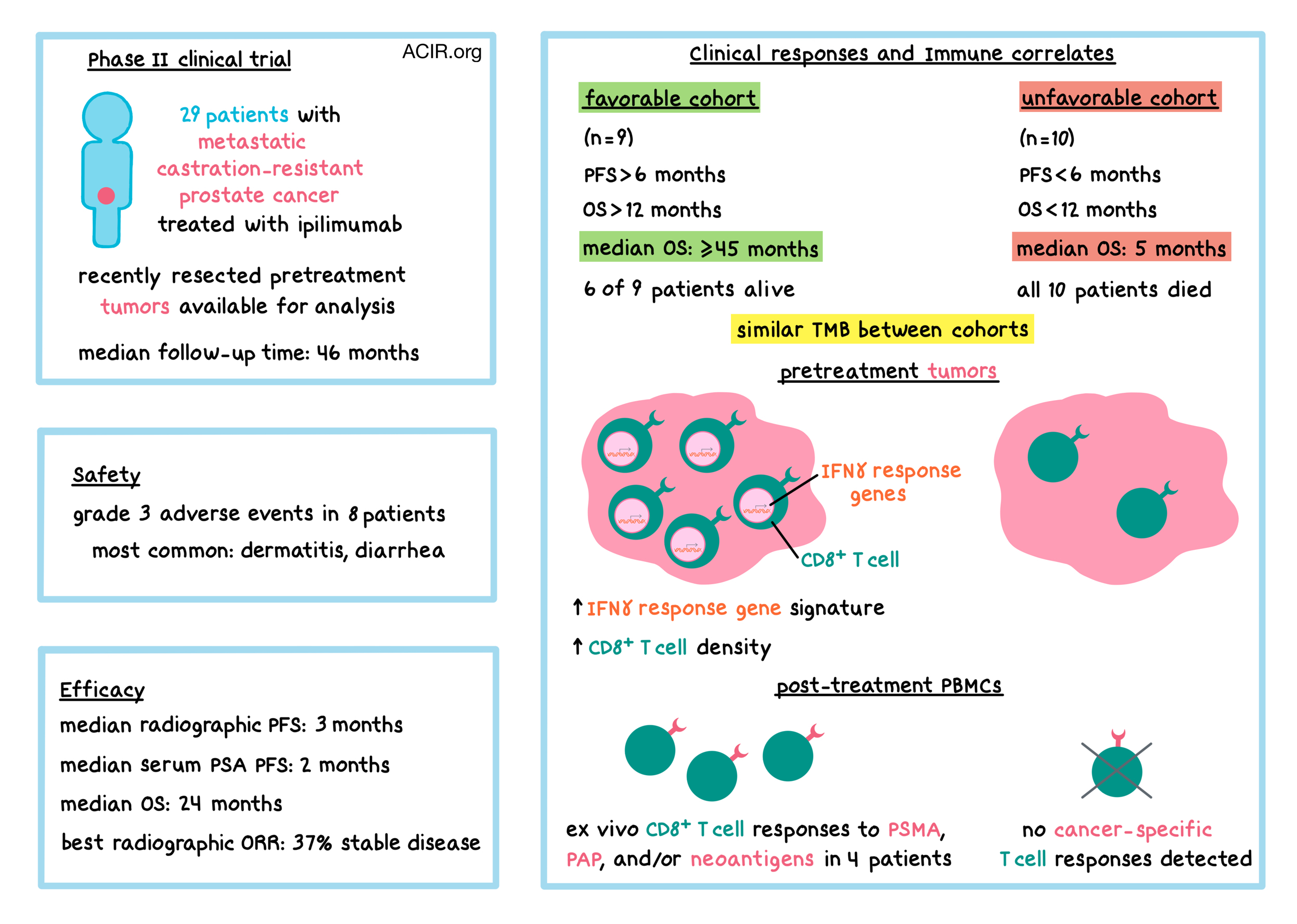
Clinical development of anticancer drugs can be enhanced using efficacy data of small population clinical trials - Sawachi - - Journal of Clinical Pharmacy and Therapeutics - Wiley Online Library
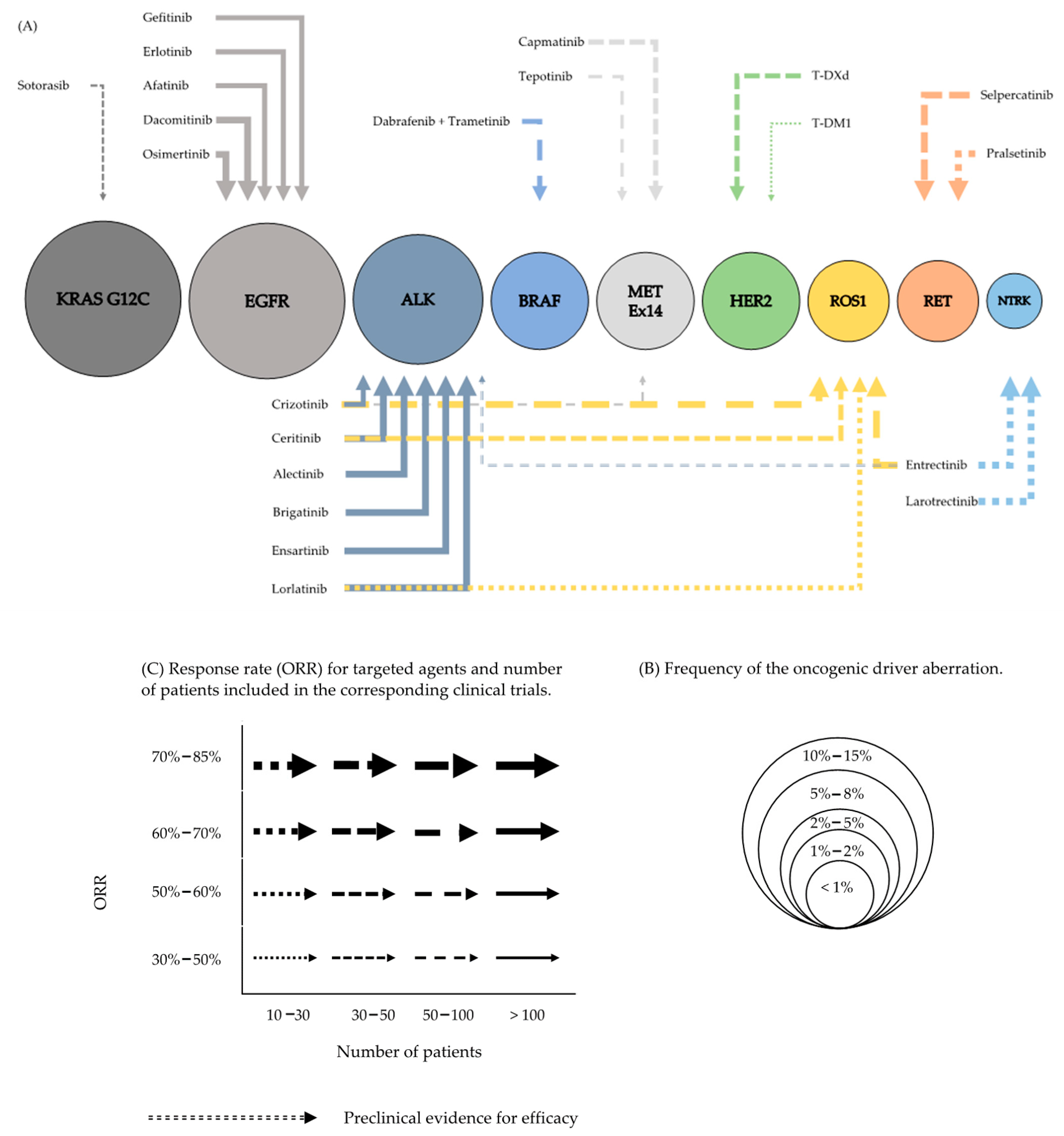
Cancers | Free Full-Text | Targeted Therapy in Advanced and Metastatic Non-Small Cell Lung Cancer. An Update on Treatment of the Most Important Actionable Oncogenic Driver Alterations | HTML

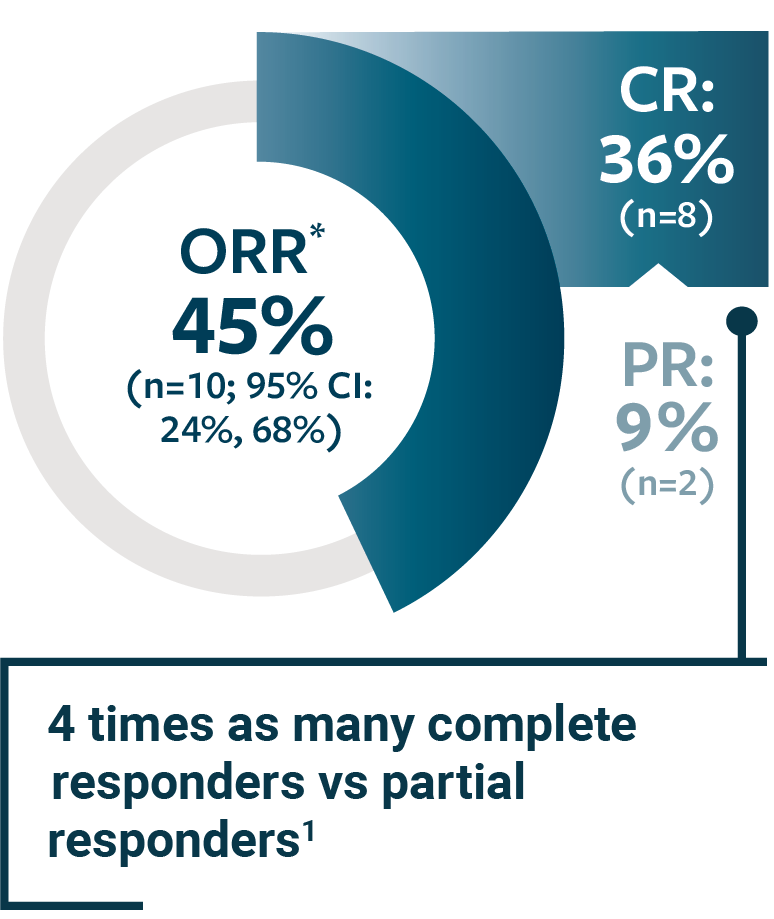
Abbreviations: CR, complete response; ORR, objective response rate; PR,... | Download Scientific Diagram
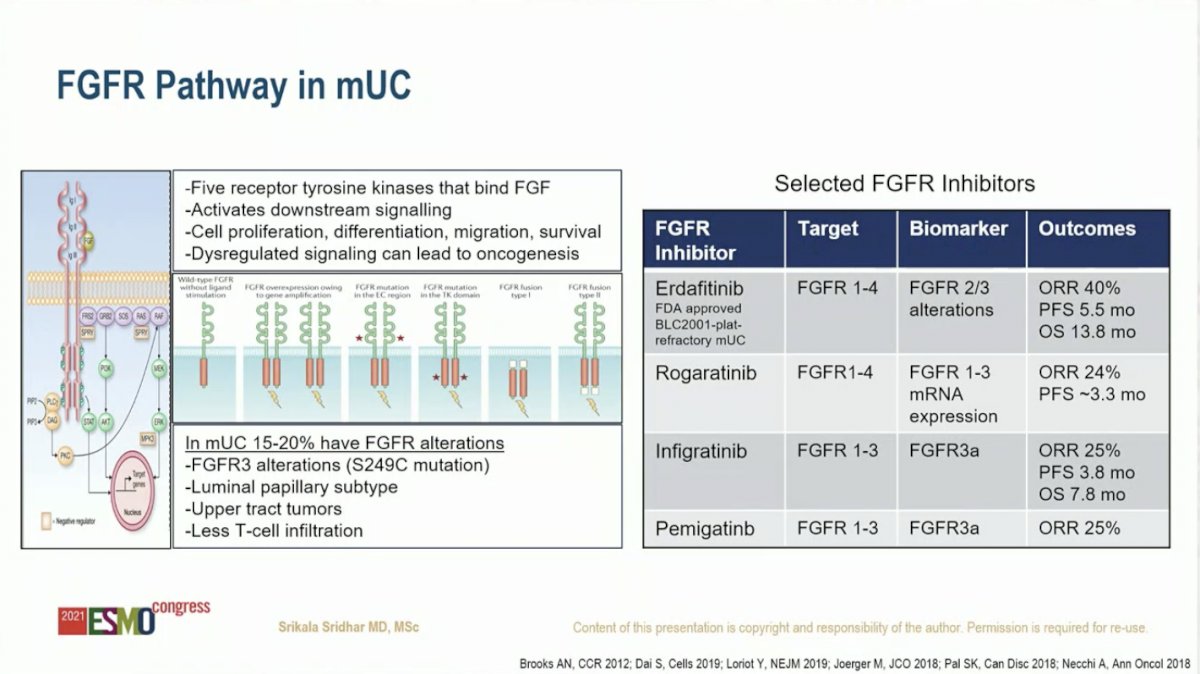
ESMO 2021: The Phase 2 NORSE Trial Discussion: A Novel Combination in the First-Line Setting for Patients With Metastatic or Locally Advanced Urothelial Carcinoma and FGFR Alterations

Risks and benefits of anticancer drugs in advanced cancer patients: A systematic review and meta-analysis - eClinicalMedicine

Phase I trials as valid therapeutic options for patients with cancer | Nature Reviews Clinical Oncology