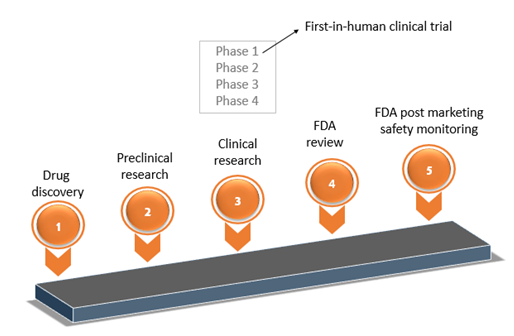
Design and Conduct Considerations for First‐in‐Human Trials - Shen - 2019 - Clinical and Translational Science - Wiley Online Library

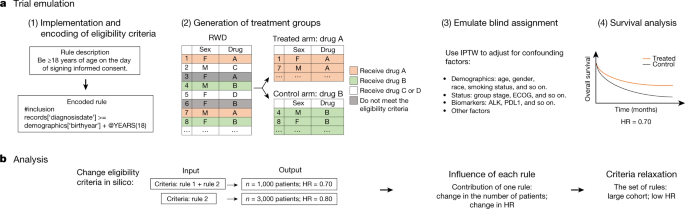
Need for aligning the definition and reporting of cytokine release syndrome (CRS) in immuno-oncology clinical trials - Cytotherapy
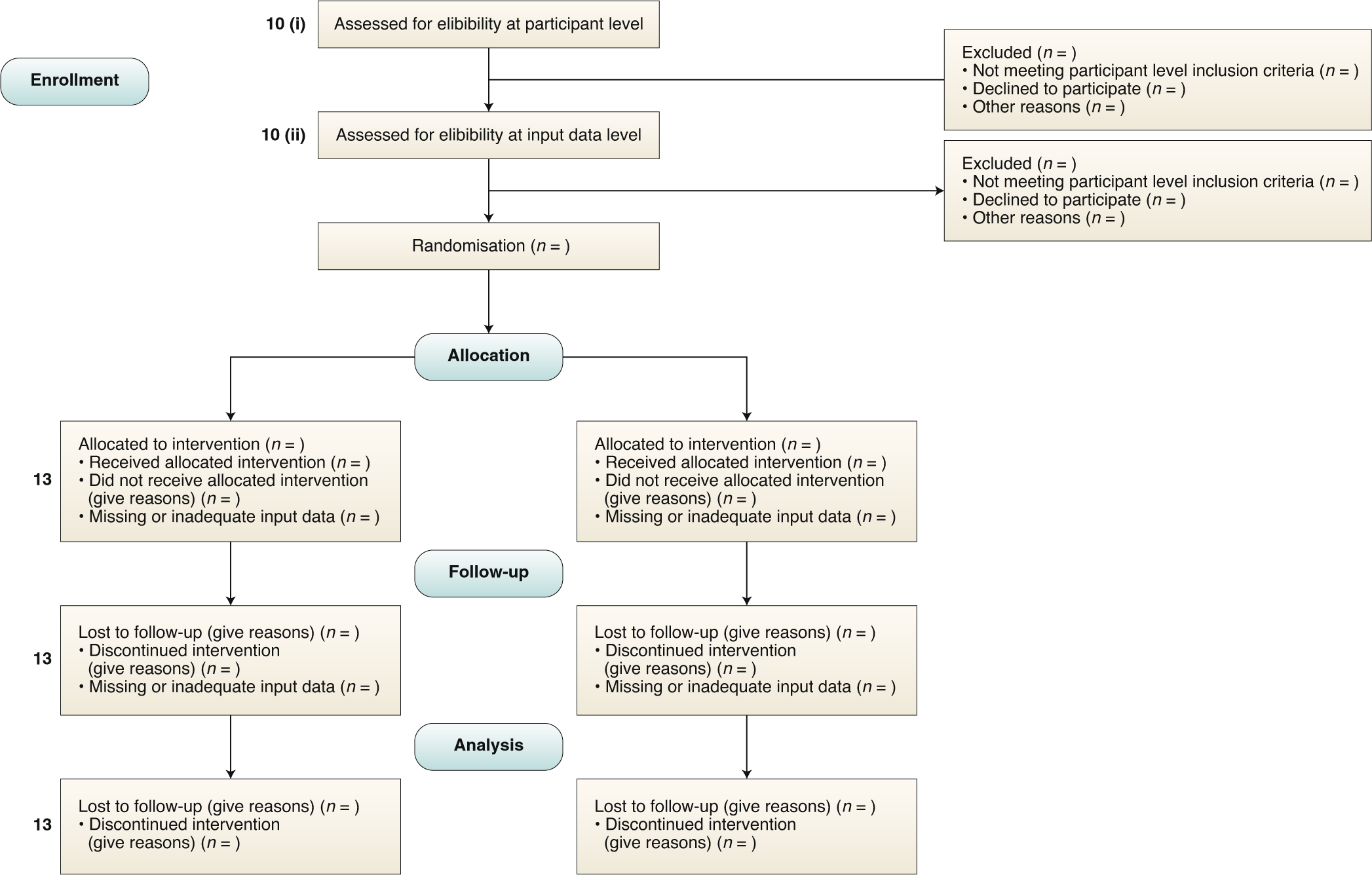
Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension | Nature Medicine

Harmonization of U.S., European Union, and Canadian First-in-Human Regulatory Requirements for Radiopharmaceuticals: Is This Possible? | Journal of Nuclear Medicine

FDA draft guidance aims to expedite first-in-human clinical trials for oncology drugs and biologics - Pearl Pathways
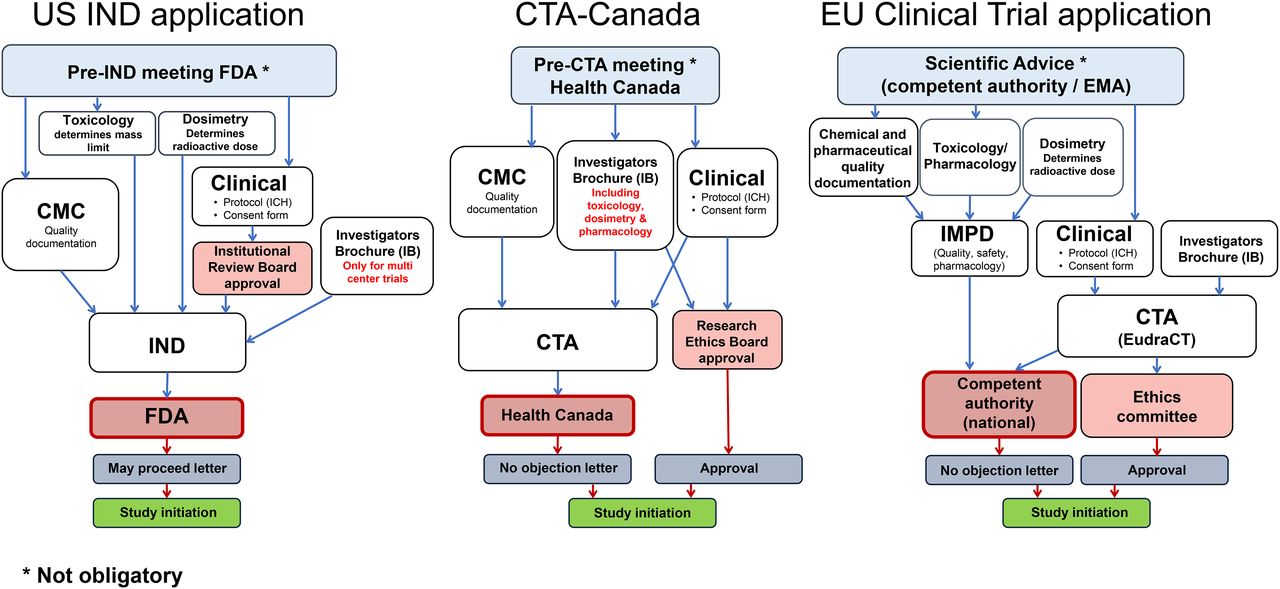
Harmonization of U.S., European Union, and Canadian First-in-Human Regulatory Requirements for Radiopharmaceuticals: Is This Possible? | Journal of Nuclear Medicine

EMA publishes revised guidelines to reduce risk in first-in-human clinical trials - The Pharmaceutical Journal